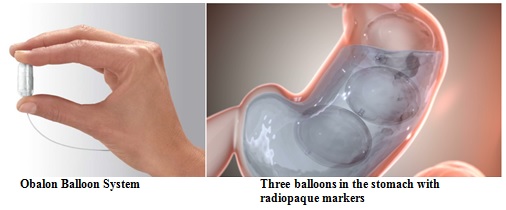
The Obalon Balloon System, a weight-loss system that uses gastric balloons to occupy space in the stomach, has been approved by the FDA for product marketing.
The Obalon Balloon System is a weight-loss system that uses gastric balloons to occupy space in the stomach. The balloons are delivered to the stomach inside of a swallowable capsule that is attached to a thin (1 mm) inflation catheter.
Once in place, the capsule opens and the balloon is filled with air via the inflation catheter. Each inflated balloon occupies approximately 1 cup of volume (250 mL). Up to three (3) balloons can be placed over a 6-month treatment period. At the end of treatment, the balloons are deflated and removed with an endoscopic procedure.
The FDA has approved this product for marketing, citing restrictions that include:
The sale and distribution of this device are restricted to prescription use in accordance with 21 CFR 801.109 and under section 515(d)(1)(B)(ii) of the Federal Food, Drug, and Cosmetic Act (the act). The device is further restricted under section 515(d)(1)(B)(ii) of the act insofar as the labeling must specify the specific training or experience practitioners need in order to use the device. FDA has determined that this restriction on sale and distribution is necessary to provide reasonable assurance of the safety and effectiveness of the device. Your device is therefore a restricted device subject to the requirements in sections 502(q) and (r) of the act, in addition to the many other FDA requirements governing the manufacture, distribution, and marketing of devices.
This is a brief overview of information related to FDA’s approval to market this product. See the links below to the Summary of Safety and Effectiveness Data (SSED) and product labeling for more complete information on this product, its indications for use, and the basis for FDA’s approval.
Continue Reading Below ↓↓↓
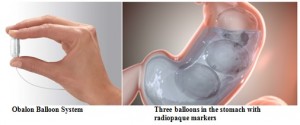
How does it work? The Obalon Balloon System occupies space in the stomach to help patients lose weight. The system is temporary and should be removed after 6 months.
When is it used? The device is used to treat adults with obesity who have a Body Mass Index (BMI) of 30-40 kg/m2 and who have been unable to lose weight through diet and exercise. The Obalon Balloon System is intended to be used while a patient participates in a moderate intensity diet and exercise program.
What will it accomplish? During the clinical study, the group of patients who used this device lost more weight than those who did not use it. The study included a total of 419 subjects, of whom 387 were able to successfully swallow the device. A total of 198 subjects received the Obalon device and 189 received a sham device (a capsule without a balloon). All study participants received diet and exercise counseling.
Patients with the Obalon Balloon System lost an average of 14.4 pounds (6.6% of their total body weight). The patients who received the sham device lost an average of 7.4 pounds (3.42% of their total body weight).
When should it not be used?
The Obalon Balloon System should not be used in patients who:
- have anatomical abnormalities or functional disorders that may affect swallowing.
- have had prior surgeries that may have led to a narrowing of the gastrointestinal tract.
- have had any bariatric surgery procedure.
- have inflammatory and other pathophysiological conditions of the GI tract.
- take medications known to be gastric irritants or those that alter function or integrity of any portion of the GI tract, including NSAIDs and aspirin.
- have an untreated Helicobacter pylori infection.
- are unable or unwilling to take prescribed proton pump inhibitor medication for the duration of the device implant.
- have allergies to products/foods of porcine origin.
- have bulimia, binge eating, compulsive overeating, high liquid calorie intake habits or similar eating related psychological disorders.
- have a history of structural or functional disorders of the stomach including, gastroparesis, gastric ulcer, chronic gastritis, gastric varices, hiatal hernia (> 2 cm), cancer or any other disorder of the stomach.
- require the use of anti-platelet drugs or other agents affecting the normal clotting of blood.
- are pregnant or lactating, or women with an intention to become pregnant.
- have a history of any obstructive disorder of the gastrointestinal tract.
- have a history of irritable bowel syndrome, radiation enteritis, or other inflammatory bowel disease, such as Crohn’s disease.
- are taking medications on specified hourly intervals that may be affected by changes in gastric emptying, such as anti-seizure or anti-arrhythmic medications.
- have untreated or unstable alcohol or illicit drug addiction.
Product Name: Obalon Balloon System
PMA Applicant: Obalon Therapeutics, Inc.
Address: 5421 Avenida Encinas, Suite F, Carlsbad, CA 92008
Approval Date: September 8, 2016
Approval Letter: PDF version
Additional information (including warnings, precautions, and adverse events): Summary of Safety and Effectiveness Data and labeling are available online:
Source: FDA