Novo Nordisk Inc. issued a voluntary nationwide recall of GlucaGen HypoKit due to detached needles on the syringe in the kit.
Novo Nordisk Inc. is recalling six batches of the GlucaGen® HypoKit® in the U.S. due to two customer complaints from the UK and Portugal involving detached needles on the syringe with Sterile Water for Injection (SWFI).
GlucaGen® HypoKit® is indicated for the treatment of severe hypoglycemia (low blood sugar) in patients with diabetes who are treated with insulin. A syringe with a detached needle cannot be used as prescribed.
Untreated hypoglycemia can eventually lead to unconsciousness and seizures, which can prove fatal. If the blood glucose levels are not quickly restored, continuing hypoglycemia can lead to a decline in brain glucose levels which manifests through a variety of symptoms including cognitive dysfunction, sweating, tremors, convulsion and eventually coma or death.
Novo Nordisk conducted an investigation which showed that a small number (0.006%) of needles could be detached from the syringe in certain batches of GlucaGen® HypoKit®. To protect patient safety, Novo Nordisk is recalling affected batches from wholesalers, pharmacies and patients in the U.S. It is estimated that out of the 71,215 pens being recalled, four pens could be defective.
This recall includes GlucaGen HypoKit batch numbers:
Continue Reading Below ↓↓↓
- Batch: FS6X270, Expiry: 09/30/2017
- Batch: FS6X296, Expiry: 09/30/2017
- Batch: FS6X538, Expiry: 09/30/2017
- Batch: FS6X597, Expiry: 09/30/2017
- Batch: FS6X797, Expiry: 09/30/2017
- Batch: FS6X875, Expiry: 09/30/2017
The affected products were distributed starting February 15, 2016.
Novo Nordisk is working as quickly as possible and in collaboration with the U.S. Food and Drug Administration (FDA) to recall affected products from the marketplace, including those in the possession of patients.
To date, Novo Nordisk is not aware of any known adverse events resulting from the use of the recalled batches.
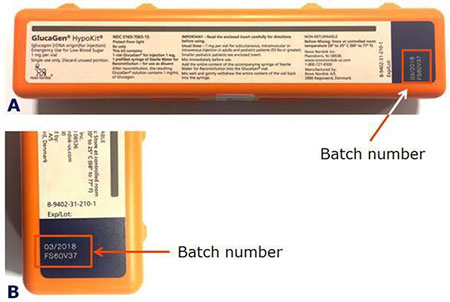
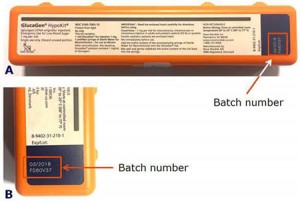
Patients or caregivers should check the batch number to see if their GlucaGen® HypoKit® is affected. The batch number is printed on the GlucaGen® HypoKit® as indicated below in the red box (Figure 1).
Figure 1. A) GlucaGen HypoKit where the batch number is found in the red box, B) close up of the batch number.
If you have a GlucaGen HypoKit® with one of the above-mentioned batch numbers, call 1-888-840-1137 from Monday to Friday, between 8:30am – 6:00pm Eastern Time, to find out how to return the product.
Novo Nordisk will provide reimbursement for out-of-pocket costs incurred for the purchase for your affected GlucaGen® HypoKit® with proof of purchase. If you received a GlucaGen® HypoKit® through the Novo Nordisk Patient Assistance Program, you will receive a replacement device.
If you are in possession of a GlucaGen® HypoKit® with a batch number NOT mentioned above, the product is not subject to the recall and may be used as prescribed.
Novo Nordisk Inc. is notifying its distributors and customers by letter and phone and is arranging for return of all recalled products.
Adverse reactions or quality problems experienced with the use of this product may be reported to Novo Nordisk by calling 1-800-727-6500. Patients can also call the FDA’s MedWatch Adverse Event Reporting program.
Continue Reading Below ↓↓↓
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
Novo Nordisk is committed to patient safety and delivering high-quality medicines, and is working to take corrective actions and avoid re-occurrence.
Source: FDA