March 2006 - Islet cell xenotransplantation presents a promising near-term solution to the critically low islet cell supply for humans suffering from type 1 diabetes, according to researchers from the Emory Transplant Center, the Yerkes National Primate Research Center of Emory University and the University of Alberta, Canada.
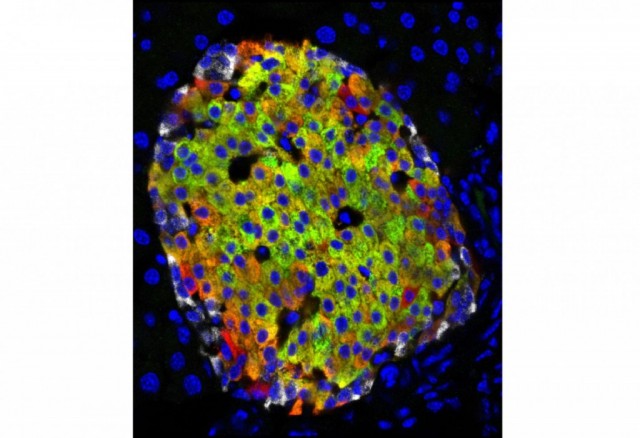
The Emory/Yerkes researchers successfully transplanted and engrafted insulin-producing neonatal porcine islet cells harvested by the University of Alberta researchers into diabetic rhesus macaque monkeys, restoring the monkeys' glucose control and resulting in sustained insulin independence.
This research, published in the February 26 advanced online edition of Nature Medicine, also examines the effectiveness of a costimulation blockade-based regimen developed at Emory proven to have fewer toxic side effects than currently used immunosuppressive regimens, and provides essential answers to the possibility of cross-species viral transmission, a common concern of xenotransplantation use in humans.
Islet cell transplantation has been successful in reversing type 1 diabetes in humans, but the limited availability of islet cells greatly diminishes the possibility of meeting the medical needs of more than one million Americans who have the disease. Each year, only 3,000 to 4,000 donor organs are available, and each organ can only produce enough cells for, at most, one transplant.
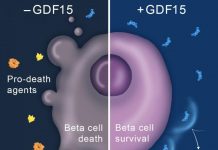
"To meet the needs of millions of people suffering from type 1 diabetes, we must find new donor sources to allow large-scale application of islet cell transplantation in humans," said Christian P. Larsen, MD, DPhil, director of the Emory Transplant Center and affiliate scientist at the Yerkes Research Center. "While there is much work to be done, these studies suggest the rejection response to porcine islets can be surmounted."
Continue Reading Below ↓↓↓
While the Emory/University of Alberta findings are similar to the recently publicized research conducted by the University of Minnesota, the nonhuman primates in this study exhibited improved glucose control and sustained insulin independence using a simpler, less toxic CD28/CD154 costimulation blockade-based regimen developed by Dr. Larsen and Kenneth Cardona, MD, of the Emory Transplant Center and the Yerkes Research Center. Belatacept, a key ingredient in the costimulation blockade regimen that selectively blocks the second of two cellular signals (costimulatory signals) the body needs to trigger an immune response, was developed between investigators at Bristol Myers-Squibb Pharmaceutical Research Institute and by Dr. Larsen and Thomas Pearson, MD, DPhil, of the Emory Transplant Center. The costimulation blockade regimen used in this study is less complex than the immunosuppressant compounds used in previous research, and its simplicity will prove more applicable in clinical use, according to Dr. Cardona.
In addition, researchers addressed concerns of the possibility of cross-species disease transmission as a result of xenotransplantation. "After extensive testing, there was no evidence of transmission of porcine endogenous retroviruses between the porcine cells and the transplant animals," said Dr. Cardona.
"The use of nonhuman primates was critical for testing cross-species viral transmission due to their close genetic link to humans. These findings are promising and suggest porcine islet xenotransplantation may prove a safe method for reversing human type 1 diabetes."
The neonatal porcine cells used in this study were harvested using a novel technique developed by Gregory S. Korbutt, MD, and Ray V. Rajotte, MD, of the Surgical-Medical Research Institute at the University of Alberta. "The harvesting method is both simple and reproducible, but the significant advantage of the technique is that the neonatal islet cells retain their growth potential post-transplant," said Dr. Rajotte, founder and director of the University of Alberta's Islet Transplantation Group. "The next steps are to prove porcine islet cells can be a source for human transplantation and to verify the safety of the transplant procedures. It's hoped within the next three to five years, we will begin testing neonatal porcine islet transplants in human patients."
Research was supported by the National Institutes of Health (NIH), the Juvenile Diabetes Research Foundation Center, Yerkes Research Center Base Grant P51-RR000165-45, the McKelvey Lung Transplant Center and the Carlos and Marguerite Mason Trust. Additional support was provided by the Canadian Institutes of Health Research, Edmonton Civic Employees Charitable Assistance Fund, Alberta Diabetes Foundation, Canadian Diabetes Association and University of Alberta Hospital Foundation MacLachlan Fund.
The Emory Transplant Center is one of the most advanced and comprehensive transplant centers in the Southeast. The center brings together Emory's transplantation programs in heart, lung, liver, kidney, pancreas and islet. The center is committed to providing and improving care and access to those in need of organ transplantation through full-service patient care and support services, as well as groundbreaking research to further the field of transplantation.
The Yerkes National Primate Research Center of Emory University is one of eight national primate research centers funded by the NIH. The Yerkes Research Center is a recognized leader for its biomedical and behavioral studies with nonhuman primates, which provide a critical link between research with small laboratory animals and the clinical trials performed in humans. Yerkes researchers are on the forefront of developing vaccines for AIDS and malaria, and treatments for cocaine addiction and Parkinson's disease. Yerkes researchers also are leading programs to better understand the aging process, pioneer organ transplant procedures and provide safer drugs to organ transplant recipients, determine the behavioral effects of hormone replacement therapy, prevent early onset vision disorders and shed light on human behavioral evolution.
Source: Emory University Health Sciences Center