A study suggests a new way to prevent vision loss and diabetic retinopathy with minimal side effects – a discovery which could affect as many as 93 million people worldwide.
Researchers at Bascom Palmer Eye Institute, part of the University of Miami Miller School of Medicine, have identified a new molecule that induces the formation of abnormal blood vessels in the eyes of diabetic test subjects.

The study, “Secretogranin III as a disease-associated ligand for antiangiogenic therapy of diabetic retinopathy,” was published March 22nd in The Journal of Experimental Medicine.
The study suggests that inhibiting this molecule may prevent similarly aberrant blood vessels from damaging the vision of not only diabetics, but also premature infants.
Changes in the vasculature of diabetes patients can cause long-term complications such as diabetic retinopathy, which affects around 93 million people worldwide.
Continue Reading Below ↓↓↓
Many of these patients suffer a dramatic loss of vision as the blood vessels supplying the retina become leaky and new, abnormal blood vessels are formed to replace them.
A molecule called vascular endothelial growth factor (VEGF) regulates blood vessel growth and leakiness, and two VEGF inhibitors, ranibizumab (Lucentis) and aflibercept (Eylea), have been approved to treat retinal vascular leakage, though they are only successful in about a third of patients.
The growth of abnormal new blood vessels also causes retinopathy of prematurity (ROP), the most common cause of vision loss in children that affects up to 16,000 premature infants per year in the US. VEGF inhibitors are not approved for use in these patients because VEGF is crucial for vascular development in newborn children.
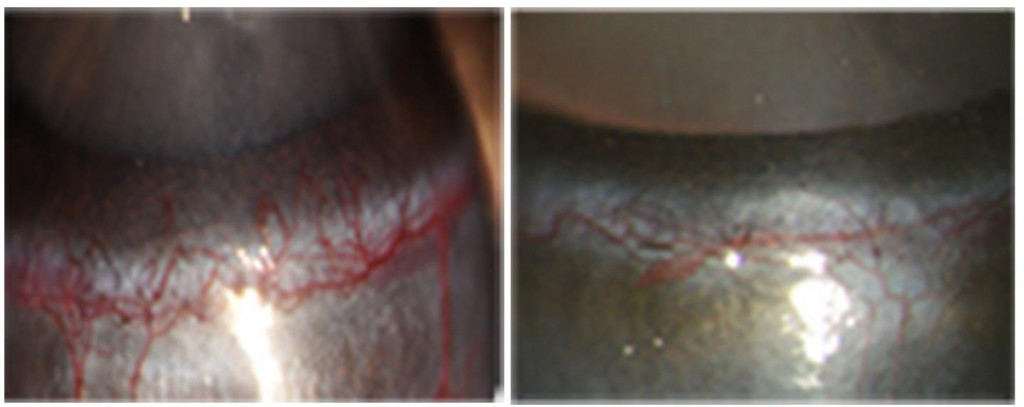
Study lead-author Wei Li, Ph.D., research associate professor, and his colleagues at Bascom Palmer developed a technique called “comparative ligandomics” to identify additional molecules that regulate the behavior of blood vessels in diabetic mice.
The approach allows the researchers to compare the signaling molecules that selectively bind to the surface of retinal blood vessel cells in diabetic but not healthy animals.
“It is estimated that between one third and one half of all marketed drugs act by binding to cell surface signaling molecules or their receptors,” says Li. “Our ligandomics approach can be applied to any type of cell or disease to efficiently identify signaling molecules with pathogenic roles and therapeutic potential.”
Using this technique, Li and colleagues discovered that a protein called secretogranin III (Scg3) efficiently binds to the surface of retinal blood vessel cells in diabetic, but not healthy, mice. Though Scg3 promotes the secretion of hormones and other signaling factors, it wasn’t thought to have a signaling function itself.
Nevertheless, the researchers found that Scg3 increased vascular leakage, and, when administered to mice, it stimulated blood vessel growth in diabetic, but not healthy, animals.
VEGF, in contrast, stimulates blood vessel growth in both diabetic and healthy mice. Li and colleagues think that Scg3 binds to a distinct cell surface receptor that is specifically up-regulated in diabetes.
Treating diabetic mice with Scg3-neutralizing antibodies dramatically reduced the leakiness of their retinal blood vessels.
Continue Reading Below ↓↓↓
Moreover, the antibodies significantly inhibited the growth of new blood vessels in mice with oxygen-induced retinopathy, a well-established animal model of human ROP.
Though the researchers still need to confirm the role of Scg3 in humans, inhibiting this protein could be an effective treatment for both diabetic retinopathy and ROP, especially as it appears to have no role in normal vascular development.
“Scg3 inhibitors may offer advantages such as disease selectivity, high efficacy, and minimal side effects,” Li says. “Because they target a distinct signaling pathway, anti-Scg3 therapies could be used in combination with, or as an alternative to, VEGF inhibitors.”
The Journal of Experimental Medicine (JEM) features peer-reviewed research on immunology, cancer biology, stem cell biology, microbial pathogenesis, vascular biology, and neurobiology.
Source: Rockefeller University Press
Photo Credit: LeBlanc et al. 2017. J. Exp. Med. http://jem.rupress.org/cgi/doi/10.1084/jem.20161802?PR
Journal: Journal of Experimental Medicine
Funders: National Institutes of Health, BrightFocus Foundation, Research to Prevent Blindness, American Heart Association