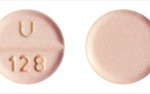
Nationwide Recall of Hydrochlorothiazide Tablets Used for Hypertension and Edema
Unichem is recalling 25mg Hydrochlorothiazide Tablets after a report that a Clopidogrel tablet was found in a bottle of the product. It can be...
Barber Foods Recalls Stuffed Chicken Products Due To Possible Salmonella
Add Barber Foods to the list of brands recalling frozen, raw stuffed chicken products that may be contaminated with Salmonella Enteritidis. Approximately 1,707,494 pounds...
Massive Recall: Frozen, Raw, Stuffed & Breaded Chicken Products
Approximately 1,978,680 pounds of frozen, raw, stuffed and breaded chicken product that may be contaminated with Salmonella Enteritidis. This recall involves almost 2-dozen brands,...
Recall: Advanced Joint Formula Capsules by G&C Natural Nutrition
G&C Natural Nutrition is voluntarily recalling all lots of it's Advanced Joint Formula capsules due to two undeclared drug ingredients - diclofenac and chlorpheniramine. Consumer...
Supplement Recall: Smart Lipo Recalled due to Undeclared Sibutramine, Desmethylsibutramine, and Phenolphthalein
122 lots of Smart Lipo are being recalling due to potentially life-threatening reactions which are especially dangerous to people with high blood pressure and...
FDA Warning: SGLT2 Inhibitors for Diabetes May Lead to a Serious Condition
The FDA is warning people about a new class of diabetes drugs that could result in ketoacidosis and may require hospitalization. The U.S. Food and...
Mucinex Recall: Several Cold Medicine Products Have Incorrect Ingredient Labels
The makers of Mucinex have issued a recall of liquid bottles of Mucinex Fast-Max Night Time Cold & Flu; Mucinex Fast-Max Cold & Sinus;...
Baxter Recalls Select Lots of IV Solutions
Intravenous administration of a solution containing sterile particulate matter may lead to adverse health consequences and should not be used. The lots being recalled...
Sabra Hummus Recalls Select Lots Due to Possible Listeria
If you purchased Sabra brand hummus recently, then make sure you check the label. 30,000 cases are being recalled due to possible Listeria contamination....
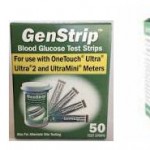
Alert for Lifescan Meter Users: Certain Blood Glucose Test Strips Reporting False Results
GenStrip Blood Glucose Test Strips, which are advertised for use with the LifeScan OneTouch family of glucose meters, may report false results. Find out...
Alert: Walnut and Trail Mix Recall Expanded Due to Salmonella
Aurora Products, Inc. is expanding its voluntary nationwide recall of certain lots of NATURAL WALNUTS and TRAIL MIXES CONTAINING WALNUTS, because they have the...
Spinach Recall Expands. Includes Wegmans, Cadia, Meijer, Target, Amy’s and more.
Due to possible Listeria monocytogenes contamination, a number of products from major stores and brands are involved in this spinach recall. This includes frozen...
Wegmans Frozen Spinach Recall, Possible Listeria Monocytogenes Contamination
Wegmans has announced a recall of frozen Wegmans Organic Food You Feel Good About Just Picked Spinach, 12 oz. which may be contaminated with...
Recall of Ultra ZX Weight Loss Supplements for Undeclared Sibutramine and Phenolphthalein
UltraZx, Labs, L.L.C. is voluntarily recalling “UltraZx” weight loss supplements. This product has been found to contain undeclared Sibutramine and phenolphthalein. FDA laboratory analysis of...
Recall of Colistimethate for Injection USP and Rifampin for Injection USP
Eatontown, NJ, Heritage Pharmaceuticals Inc. (Heritage) today announced the voluntary nationwide recall of ten (10) lots of Colistimethate for Injection, USP, 150 mg Single-Dose...
Recall of Certain Older Heartware Clinical Trial Controllers
Framingham, Mass. — HeartWare International, Inc. (Nasdaq: HTWR), is issuing a voluntary Urgent Medical Device Recall in the United States related to older HeartWare®...