A new study shows promising progress for a new treatment of type 1 diabetes using a protein made by immune cells, which essentially cured the disease.
New research from Uppsala University shows promising progress in the use of anti-inflammatory cytokine for treatment of type 1 diabetes.
The study, published in the open access journal Scientific Reports (Nature Publishing Group), reveals that administration of interleukin-35 (a protein made by immune cells) to mice with type 1 diabetes, reverses or cures the disease by maintaining a normal blood glucose level and the immune tolerance.
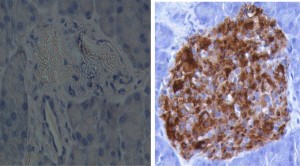
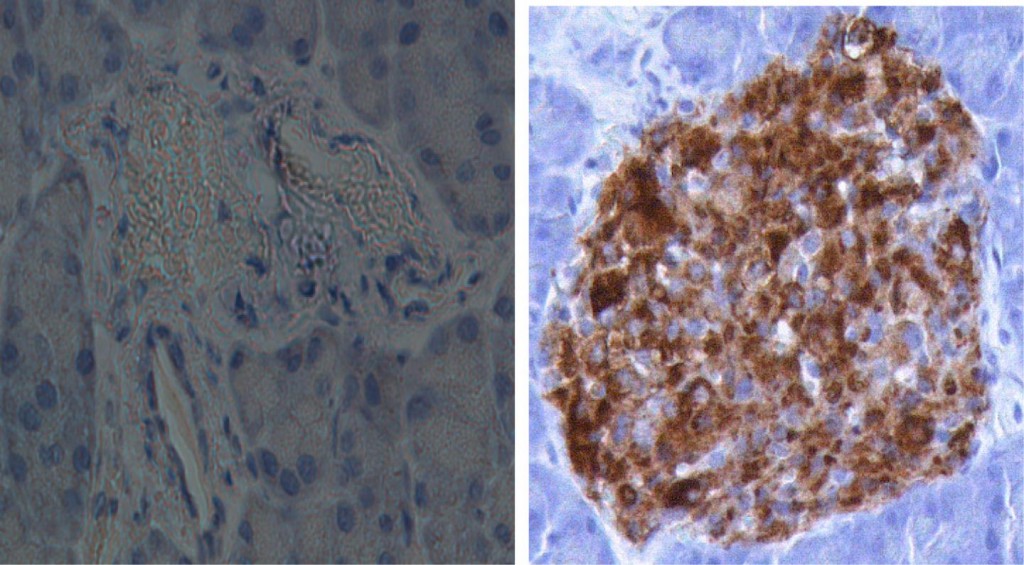
Type 1 diabetes (T1D) is a chronic disease, which for the patients leads to a life-long dependence of daily injections of insulin. In Sweden approximately 2 new cases of the disease are diagnosed every day. Insulin is a hormone, which is produced by the beta cells in the pancreas. Insulin is required to prevent a harmful rise in the blood glucose level.
Continue Reading Below ↓↓↓
The exact cause of T1D is not yet known, however, it is considered as an autoimmune disease. A condition that occurs when our own immune system by mistake attack and destroy healthy cells. In T1D, an infection and/or unknown factors probably triggers the immune cell attack, which ultimately leads to an insufficient insulin production.
In the new study, Dr. Kailash Singh, a PhD student in professor Stellan Sandler’s research group at the Department of Medical Cell Biology at Uppsala University, studied so-called immune regulatory T cells’ actions in T1D mouse models.
The study shows that the immune regulatory T cells alter their function by producing pro-inflammatory destructive proteins instead of protective anti-inflammatory proteins such as interleukin-35 (IL-35) under T1D conditions.
“This suggests that the good guys have gone bad in early development of Type 1 diabetes and therefore our immune cells destroy the beta cell”, says Dr. Kailash Singh.
Furthermore, the concentration of IL-35 was lower in T1D patients compared to healthy individuals. These findings may suggest that IL-35 could play a crucial role in human T1D.
In addition, the researchers have found a novel mechanism that explains how the immune regulatory T cells are changing their destiny under a T1D condition.
Professor Sandler’s research team tested whether or not IL-35 could also suppress development of T1D and reverse established T1D.
To induce T1D in mice they injected a chemical compound called streptozotocin. These mice developed signs of TID and increasing blood glucose levels similar as in human T1D.
IL-35 injections given after disease induction prevented from development of T1D. Strikingly, IL-35 injections to mice, which were diabetic for two consecutive days, normalized blood glucose concentrations.
The research team also successfully investigated IL-35 in another model of T1D called non-obese diabetic mouse (NOD). The interruption of IL-35 treatment did not result in return of diabetes in any of the mouse models.
Continue Reading Below ↓↓↓
The findings encourage further research on the use of IL-35 for treatment of T1D and offer new clues as to why immune regulatory T cells fail in counteracting T1D.
“To the best of our knowledge, we are the first to show that IL-35 can reverse established Type 1 diabetes in two different mouse models and that the concentration of the particular cytokine is lower in Type 1 diabetes patients than in healthy individuals. Also, we are providing an insight into a novel mechanism: how immune regulatory T cells change their fate under autoimmune conditions”, says Dr. Kailash Singh.
The research has been led by Professor Stellan Sandler, Dr. Kailash Singh and Dr. Lina Thorvaldson in collaboration with Professor Per-Ola Carlsson and Dr. Daniel Espes at the Department of Medical Cell Biology, Uppsala University, Sweden.
Publication: Singh, K. et al. Interleukin-35 administration counteracts established murine type1 diabetes – possible involvement of regulatory T cells. Sci. Rep. 5, 12633; DOI: 10.1038/srep12633 (2015).
Source: Uppsala University
Journal: Scientific Reports
Photo Credit: Kailash Singh









Sign me up for a participation!