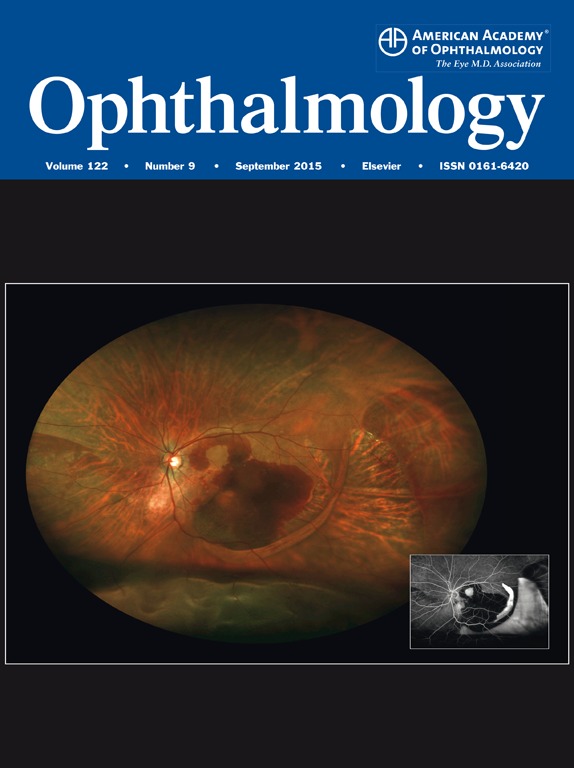
Damage in retinal periphery closely matches loss of blood flow in people with diabetes and a different imaging test may aid in earlier detection and treatment.
Research from the Joslin Diabetes Center’s Beetham Eye Institute demonstrated earlier this year that in people with diabetic retinopathy, the presence of lesions in the periphery of their retina substantially increases the risk that the disease will progress more rapidly.
A follow-up study has shown that these peripheral lesions, which are not detected by traditional eye imaging, correlate very closely with the loss of retinal blood flow called retinal “non-perfusion” caused by loss of small blood vessels or capillaries.
In research reported this week in Ophthalmology, the Joslin scientists used ultra-wide-field (UWF) imaging to examine the eyes of 37 patients with diabetes and varying levels of retinopathy ranging from no disease to very advanced disease.
UWF retinal imaging can view more than 80 percent of the retina in a single image. In comparison, traditional clinical retinal imaging combines seven smaller photos to cover about a third of the retina, says Paolo Silva, M.D., staff ophthalmologist and assistant chief of telemedicine at the Beetham Eye Institute.
Continue Reading Below ↓↓↓

Areas of non-perfusion were identified by UWF retinal angiography, which detects blood flow after patients are injected with a fluorescent dye.
“With the UWF angiograms, we can more accurately measure the extent of non-perfusion in the peripheral and examine how this relates to the increased risk for retinopathy progression over time,” says Silva, who is lead author on the paper.
The areas of non-perfusion matched up very closely with the peripheral lesions detected when these patients’ eyes were scanned by normal UWF imaging, he emphasizes.
“The most surprising result was how very closely associated these two areas seemed to be,” says Lloyd Paul Aiello, M.D., Ph.D., director of the Beetham Institute, professor of ophthalmology at Harvard Medical School and senior author on the paper.
He says that discovery raises the possibility that clinicians might be able to use the peripheral lesions to estimate the extent and location of the non-perfusion damage and the risk of disease progression without necessarily resorting to UWF angiography in every case.
A related trial by the Diabetic Retinopathy Clinical Research Network, now underway, will follow more than 350 diabetes patients across the United States with UWF imaging for at least four years.
If this national study confirms the Joslin findings about the association of peripheral lesions with risks of disease progression, and the close match of these lesions with non-perfusion, the grading system for diabetic eye disease probably will be revised to incorporate these risk factors and imaging approaches, Aiello says.
The Joslin team is now analyzing another alternative approach to assess levels of retinal perfusion–detecting levels of oxygen in blood cells non-invasively by measuring their absorption of a certain wavelength of light.
Aiello notes that the last decade has seen many major success stories for the treatment of another form of diabetic eye disease, diabetic macular edema, with drugs that target a protein called vascular endothelial growth factor (VEGF). It’s possible that such anti-VEGF drugs might also help to treat peripheral lesions, he speculates.
In any case, better understanding of these lesions and their causes represents “another step forward in the evaluation, risk identification and the treatment of patients with retinopathy.” Aiello says. “We have so many more effective treatment options than we had even three to four years ago.”
Continue Reading Below ↓↓↓
Joslin co-authors on the paper included Ajlan Radwan, Jerry Cavallerano, Lloyd M. Aiello and Jennifer Sun. Amanda Dela Cruz and Migil Ledesma of the Philippine Eye Research Institute and Jano van Hemert of Optos also contributed to the research. The work was supported in part by the Philippines National Academy of Science and Technology, the Amelia Peabody Charitable Fund and the Massachusetts Lions Eye Research Fund.
Source & Photo Credit: Joslin Diabetes Center
Journal: Ophthalmology